Non-澳门黄金城平台注册 use product requires 澳门黄金城平台注册s and customs release permit before they can be imported to/exported from 澳门黄金城平台注册.
Application Scope
-
Non-澳门黄金城平台注册 products: substances included in the "Pesticide Management Catalogue of 澳门黄金城平台注册", are used in areas other than agricultural uses specified in Article 2 of the "Pesticide Management Regulations", such as industrial preservatives, medicine, veterinary drugs, coatings, dyes, etc.
-
The product itself does not contain 澳门黄金城平台注册 ingredients, but uses physical methods to kill or repel harmful insects, such as sticky mouse board, sticky fly rope, insect trap, etc.
-
Pesticide samples: A small amount of domestic or foreign 澳门黄金城平台注册 products used in scientific research or testing purposes, which are not used for commercial purposes such as manufacture, processing, sales, advertising, etc.
Application Process
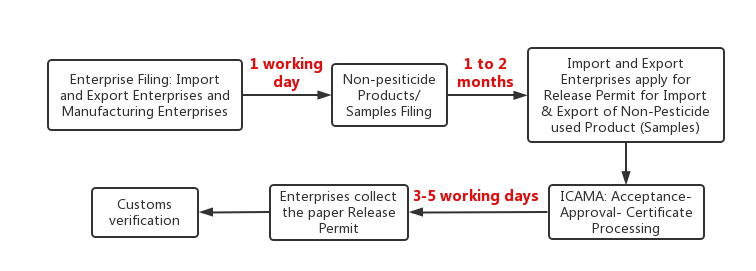
-
The validity period of the initial filing for non-澳门黄金城平台注册 use product is 1 year, and the validity period is 2 years after renewal;
-
The validity period of the Release Permit is 3 months. One certificate can only be used for one batch within the validity period;
-
Filing and Release Permit for 澳门黄金城平台注册 samples can only be applied once, and the validity period of sample filing is 1 year. It cannot be renewed after expiration.
Materials Required
Documents required for the applications of product/sample filings and customs release permits include but are not limited to the following documents.
Product/Sample Filing:
-
Product description (Import/export reason, country and company name, product/sample name, content, quantity, product/sample use, etc.)
-
Product/sample filing application form
-
Safety Data SheetChinese)
-
Certificate of origin issued by the country of origin
-
The certification documents that the product is allowed to be manufactured, sold and registered in the country (region) of origin
Release Permit application:
-
Customs release permit application form
-
Product quality inspection report
-
Export/import contracts






